
The terminal cell voltage is, therefore, the open circuit voltage (or electromotive force, EMF, if no fuel is lost by any means) reduced by the individual voltage loss of each cell component. 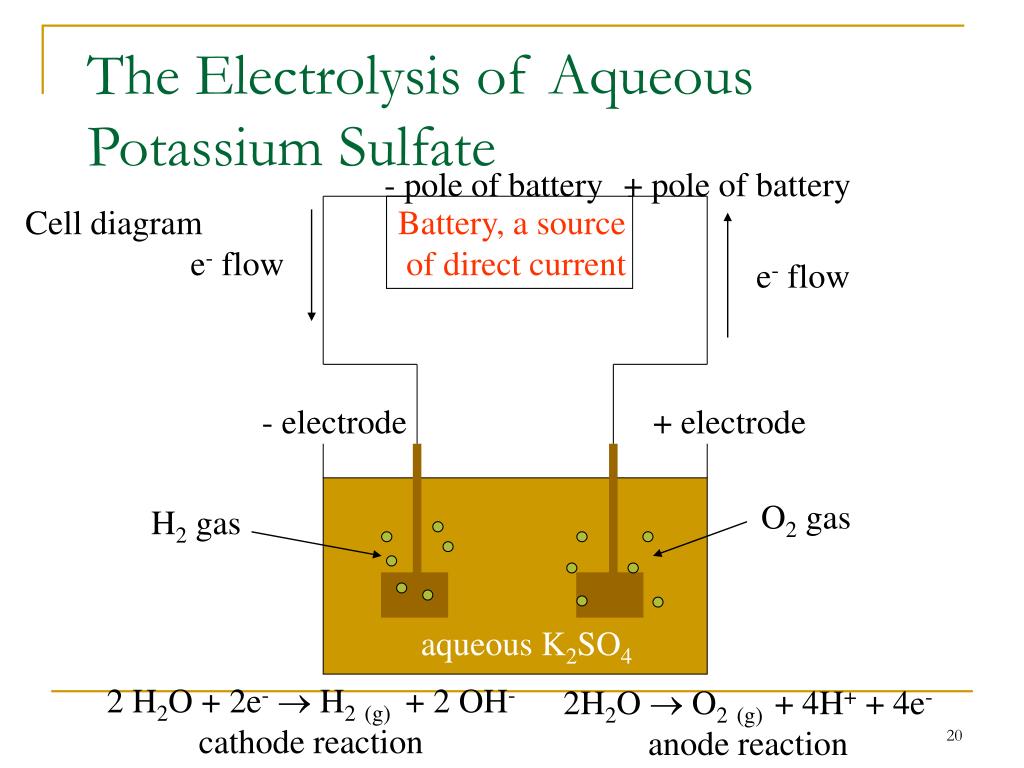
Like the components of a battery, each SOFC component exhibits an internal resistance to either electronic or ionic current flow, often expressed as voltage loss. References 1–3 provide a more detailed review of SOFC technology. The overall driving force for a SOFC is the gradient of oxygen chemical potential that exists between the cathode with a high oxygen partial pressure and the anode with a low oxygen partial pressure. The electrons required for the cathode reaction are released by the anode and arrive at the cathode via an external load that produces electricity. At the anode, or fuel supply electrode, the O 2– delivered by the electrolyte reacts with hydrogen or a hydrocarbon fuel to produce H 2O, CO 2, and electrons. The electrolyte transports oxygen continuously, in the form of O 2–, from the cathode to the anode under a gradient of oxygen chemical potential. The cathode reduces oxygen (in the form of O 2) in the air supplied to it into O 2–. A SOFC ( Figure 1) has three basic functional elements: cathode, electrolyte, and anode. 
In a solid oxide fuel cell, the cathode reduces oxygen (from air) to O 2–, which the electrolyte transports from the cathode to the anode, where it reacts with a fuel to produce H 2O, CO 2, and electrons.Ī solid oxide fuel cell (SOFC) directly converts the chemical energy in fossil fuels into electrical power via an electrochemical reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
